This article first appeared in The Edge Malaysia Weekly on October 12, 2020 - October 18, 2020
IT is not a widely known fact that Penang, famous for its thriving electrical and electronics (E&E) industry, also has a burgeoning medical device industry. The state is slowly gaining a reputation as a medical device manufacturing hub among world-class manufacturers.
Its most recent investments include US company DexCom Inc, a global leader in continuous glucose monitoring, and Switzerland-based electrical measurement company LEM.
Malaysia is seen as the leading medical device manufacturing hub in the East, competing against the likes of mature hubs such as Puerto Rico, Costa Rica and Ireland.
While central Malaysia attracts latex-related medical device companies such as those in glove and condom manufacturing because of its proximity to raw materials, Penang is making a name for itself in non-glove medical device manufacturing or, more specifically, medical technology (medtech).
Unlike industries with low barriers to entry, medical device producers usually stay put once they have decided on a location for their manufacturing base.
Devices manufactured in Penang tend to be on the higher end of the value scale, such as cardiovascular products or orthopaedic implants and tools.
Invest-in-Penang Bhd (InvestPenang) CEO Datuk Loo Lee Lian says the sector is actually an offshoot of the state’s E&E sector. Part of the credit for its growth should be given to B Braun, she adds. It was the first foreign medical devices company to set up operations in Penang in 1972, around the same time the “8 samurais” of the E&E sector arrived here.
“B. Braun was the training ground that started the medical devices industry in Malaysia. A lot of the CEOs in the medical device industry today are alumni of B. Braun.
“They did for the medical devices industry what companies like Intel did for the semiconductor industry. B. Braun helped create the ecosystem for the medical device industry in Penang,” she explains.
The rapidly growing E&E sector in Penang created opportunities for some companies in the industry to transition into medical device manufacturing.
“We realised in 2005 that it is easier for the existing E&E and manufacturing industrial players in Penang to transition into medical device manufacturing by leveraging similar manufacturing skillsets in the E&E industry.
“Since then, the Penang state government has been focusing on growing the medical device cluster by working closely with the captains of the industry,” says Ching Choon Siong, executive director of the Association of Malaysian Medical Industries (AMMI).
Penang’s advantages
Ching says Penang’s success at attracting foreign investments is due to its competitive costs and established infrastructure. But equally important is the talent pool available, which is partly the result of the clustering effect of the medical device supply base.
T H Su, CEO of home-grown Straits Orthopaedics Manufacturing Sdn Bhd, says Penang is considered as the best-cost manufacturing hub these days, not a low-cost base like before. He explains that other factors, such as a skilled workforce and logistics infrastructure, have made up for the lower costs offered by other hubs.
Dave Mitchell, Boston Scientific vice-president and general manager for manufacturing and distribution in Malaysia, shares similar sentiments. The strong collaboration among the industries that he experienced here helped cement the company’s decision that Penang was a good place to do business.
“InvestPenang was specifically helpful in helping us network with other companies. They connected us with other companies, both in the medical devices and other high tech industries, when we were here looking. That really helped us understand how we would fit and how our needs could be met,” he says.
Boston Scientific set up a massive 375,000 sq ft manufacturing plant in Batu Kawan in 2017, its first factory in Asia.
“Besides the usual aspects of site selection like economics, the availability of talent, labour, infrastructure and also intellectual property protection — which is very important to a medtech company like us — Malaysia also has the advantage of English fluency, which helps a lot because most of our R&D teams are English-speaking,” says Mitchell.
The sector’s growth story in Penang is remarkable. Loo relates that between 2011 and 2015, annual approved investments from scientific and measuring equipment, under which medtech is categorised, was below the RM600 million mark.
In 2016, it started to see a pick-up in investments, with the number reaching RM983 million. By 2019, of the RM2.52 billion of approved investments in the scientific and measuring equipment segment in Malaysia, RM2.45 billion was from Penang.
Malaysian Investment Development Authority (MIDA) statistics show that in 1Q2020, Penang recorded RM1.85 billion out of the RM1.92 billion total approved investments for the segment.
Notably, more than 95% of the investments for this sector are foreign direct investment, evidence that the medical device industry in the country is dominated by multinational corporations (MNC).
MNC presence unmissable
Depending on how one looks at it, having multinational corporations in an industry is an advantage. In Ching’s opinion, when an MNC sets up a manufacturing site, it creates opportunities for local suppliers of raw materials, services and value-added outsourcing. In the process, these local companies can gain skills and knowledge of the sector, possibly enabling them to venture into the manufacturing business.
He likens MNCs’ presence in Penang to the concept of an anchor tenant in a shopping mall, which naturally draws others to the mall.
“The strategy makes sense for us because without them, we find ourselves unable to go into a new sector sometimes. So, we might as well bring them in here and create more business opportunities for local business suppliers. There is also a chance that those working at these MNCs will themselves become entrepreneurs eventually,” says Ching.
Based on a survey by AMMI, Ching says MNCs that are members of AMMI have sourced RM550 million worth of local materials from local suppliers, RM1.74 billion of services from local small and medium-size enterprises and RM1.92 billion worth of contract manufacturing opportunities.
Nevertheless, InvestPenang’s Loo believes that more improvements can be made to localise the supply chain but admits the process will take time. InvestPenang has been pushing the MNCs to do so but the amount of localisation has been lower when compared with the E&E sector.
“The reason for the slow progress is because of the stringent [United States Food and Drug Association] requirements and lengthy qualification process. They don’t want to have to requalify their suppliers. The parts from suppliers are small in size and since they have gross margins of 70% to 80%, they are not cost sensitive and would rather fly in their parts from existing qualified suppliers wherever they are in the world,” she explains.
Loo points out that a cluster is key for the development of the sector. The medical device industry, like the E&E industry, tends to cluster.
“When you have a cluster, you attract talent. If you build the ecosystem, the supporting industries (will grow) as well. We are growing that cluster slowly but surely.”
While MNCs dominate, there are a few home-grown companies in the space.
A notable one is Straits Orthopaedics, which is a contract manufacturer for orthopaedic implants. It is also an associate company of listed healthcare group Apex Healthcare Bhd.
Currently, more than 50% of Straits Orthopaedics’ products are supplied to medical technology company Smith & Nephew plc, which focuses on orthopaedics, sports medicine and ENT as well as advanced wound management.
Straits Orthopaedics has been a contract manufacturer for Smith & Nephew for the last 17 years.
Smith & Nephew recently started construction of a 250,000 sq ft manufacturing facility in Batu Kawan Industrial Park to support its orthopaedics franchise, which has been growing strongly in Asia Pacific.
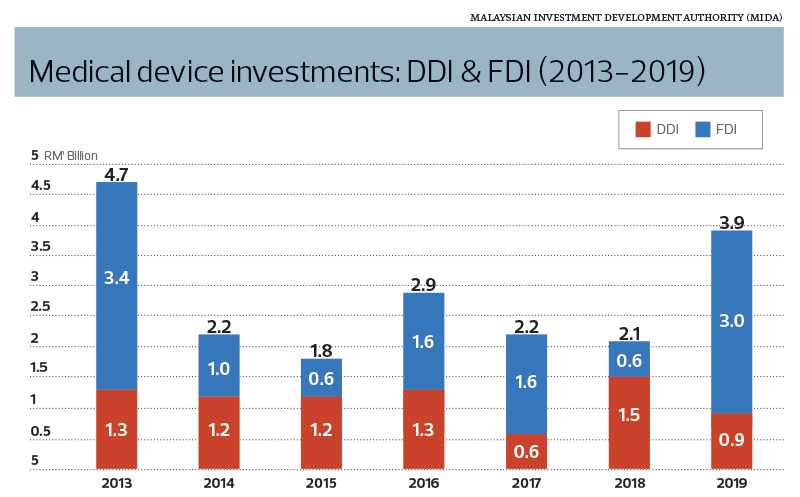
Encouragingly, Ching notes that there has been growth within the domestic industries. Between 2013 and 2019, a total of RM8 billion in domestic direct investment (DDI) was approved compared with RM11.8 billion in FDI, according to MIDA statistics.
“DDI’s share of 40% cumulative investment over this period is very encouraging,” states Ching.
The handful of local companies in the industry believe that there is room for more local players but they say more needs to be done to encourage this.
Michael Hng, managing director of Allen Healthcare Products (M) Sdn Bhd, a Malaysian company pioneering Rapid Diagnostics Test kits in Malaysia, says one of the main challenges for entry is the high investment cost.
“Some of the main factors that hinder local players from setting up a full-scale medical device manufacturing plant in Malaysia is the high investment capital, incomplete supply chain of supportive materials, regulatory requirements and compliance cost and a long seeding time before a product becomes marketable,” he adds.
Hng hopes to see more collaboration between the Ministry of Health and local medical devices manufacturers as the ministry is the biggest customer for these products.
Malaysia is the largest medical device market in Southeast Asia, with a market worth US$1.55 billion (RM6.44 billion). Public sector procurement accounts for about 50% of that, notes AMMI’s Ching.
Hng also believes that there needs to be a paradigm shift in mindset for both the government and private sector to move from merely “repackaging” to becoming full manufacturing set-ups.
Propelling the industry further
While lots of hard work and money has been invested to make Penang the medical device hub it is today, the industry believes much more can be done to keep Malaysia, specifically Penang, as the choice for world-class medical device companies.
One of the industry’s main concerns is that the types of incentives provided do not necessarily meet its needs. It is calling for incentives that are tailored for them.
Boston Scientific’s Mitchell, for one, says there needs to be improvement in the investment incentives programme, particularly in the area of reinvestments.
“For example, if, in five years, we are going to go into another significant investment, we find that other countries are better in reinvestment support. A new product can be made in any of our other 29 plants around the world. So, it is about how the country attracts reinvestment for new products,” says Mitchell.
AMMI’s Ching too believes that while the government has been very generous with grants and money for businesses, the incentives are outdated.
“Current incentives for R&D given to companies are for buying equipment. For R&D, we actually need to spend money on human capital. There is a lack of holistic incentives right now to support the R&D of medical devices,” he says.
He points out that Singapore, which emphasises R&D, is offering to pay 50% of the salaries of companies’ R&D engineers.
“The government co-invests. It is a good strategy and a better commercialisation than our current strategy here,” Ching says.
However, the local players see things a little differently.
“To me, the grants or the incentives aren’t the main things. The main thing is for regulators and authorities to assist companies in every way they can. But I feel that they don’t understand how steep the competition is like outside of Malaysia.
“Emerging countries like Vietnam have a very aggressive investment board and they have a hungry workforce of both skilled and unskilled workers. They have a huge talent pool of engineers and they also have a relatively younger population.
“They may not be ‘there’ yet, but with time, they will build up their infrastructure and they will soon close the gap,” says Straits Orthopaedics’ Su.
Says AHP’s Hng: “I don’t need the money or grants. What I would like is for regulators to support the local industry in terms of sales. We produce world-class products too.”
Besides incentives and support, B. Braun says improvement can be made with greater emphasis on the training and development of highly skilled talents in the country.
“[One way] of doing this is to instil great interest in the STEM (Science, Technology, Engineering and Mathematics) subjects while students are still in primary and secondary school. Introduce medtech content as an elective engineering course in local universities or give prominence to the medical device industry at career fairs as an industry of choice for graduates and job seekers,” says B Braun Asia Pacific president Lam Chee Hong.
AMMI’s Ching hopes that in the next five to 10 years, Malaysia will be able dominate certain sub-sectors of the medical device industry, like orthopaedics, high-value electronic-based medical products or even plastic-based products.
“In order for that to happen, we have to be good in certain products, [and] we focus our attention in these areas. Today, we are doing well in rubber glove manufacturing. In the future, we hope to see Malaysia doing well in other sectors.
“I believe the metal-related industries can do as well as the plastic-based products. So, our vision is to leverage our skill sets to do well in areas that we are strong in.”
Save by subscribing to us for your print and/or digital copy.
P/S: The Edge is also available on Apple's App Store and Android's Google Play.